Taiwan halts Pfizer second dose in ages 12 to 17 due to myocarditis risks, Not yet approved for children aged 5 -11, Risk 10 times higher than after the first dose
“it is universally known that children virtually never die from COVID-19 and given that children have a very strong immune system, they are more likely than adults to have an over-reaction to the shot.”… Dr. Steven Roth
“Because COVID vaccines use experimental technology that may pose serious side effects such as blood clots, Yeadon said, “we should absolutely not be offering them to young, healthy people who are not at risk from the virus.””..Dr. Mike Yeadon, former vice president of Pfizer
” if you let your healthy teen – much less your healthy child – get this vaccine, you are insane.”…Alex Berenson
From the Taiwan News.
“Taiwan halts 2nd-dose BioNTech vaccinations for ages 12-17 amid concerns of myocarditis
CECC says approval of COVID vaccines for children under 12 will not be considered until 2nd dose issue settled
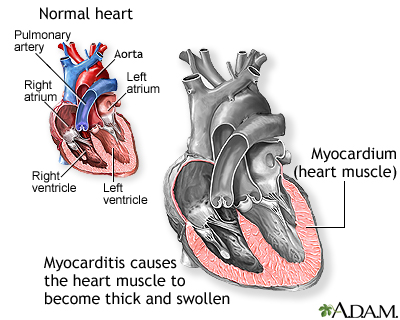
Central Epidemic Command Center (CECC) head Chen Shih-chung (陳時中) said on Wednesday (Nov. 10) that a panel of experts has decided to suspend administering second doses of the Pfizer-BioNTech (BNT) COVID vaccine to children 12-17 years old amid concerns it may increase the risk of myocarditis.
Cases of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the outer lining of the heart) have been reported after BNT vaccination of children between 12 and 17 years of age. According to U.S. statistics, the risk of youths experiencing myocarditis after receiving the second BNT dose is 10 times higher than after the first dose, CNA reported.”
“Chen said that the Ministry of Health and Welfare’s Advisory Committee for Immunization Practices (ACIP) has decided to halt administration of second BNT doses to this age group for two weeks, during which time experts and Centers for Disease Control (CDC) physicians will look at the 16 cases of myocarditis among adolescents after BNT vaccination before making a final decision on whether to go ahead with the second shot.”
“As for whether COVID-19 vaccines will be approved for children aged 5-11, Chen said the matter will not be considered until the second dose issue with 12 to 17-year-olds is settled.”
Read more:
https://www.taiwannews.com.tw/en/news/4340862
Read more here:

“Children have a greater chance of drowning, dying in a car wreck or dying from the flu than from the coronavirus.
Children are not forced to take the flu vaccine.”…GateWay Pundit